














Customized Liquid Preparation System for Biopharma with SCADA Monitoring
The Pharmaceutical Liquid Formulation System is a specialized pharmaceutical apparatus designed for liquid mixing and dispensing operations. This equipment enables the preparation of pharmaceutical solutions by combining active pharmaceutical ingredients (APIs) with solvents such as water for injection. Through integrated processes including controlled stirring, heating, sterilization, and filtration, the system produces medicinal solutions at precise target concentrations.


Pre-tested modular equipment reduces installation costs, accelerates delivery, and ensures flexibility.

1-click CIP/SIP with auto-detection for cleaning/sterilization completion. Computerized features include Recipe management, e-signature, e-records, and audit trail for full automation.
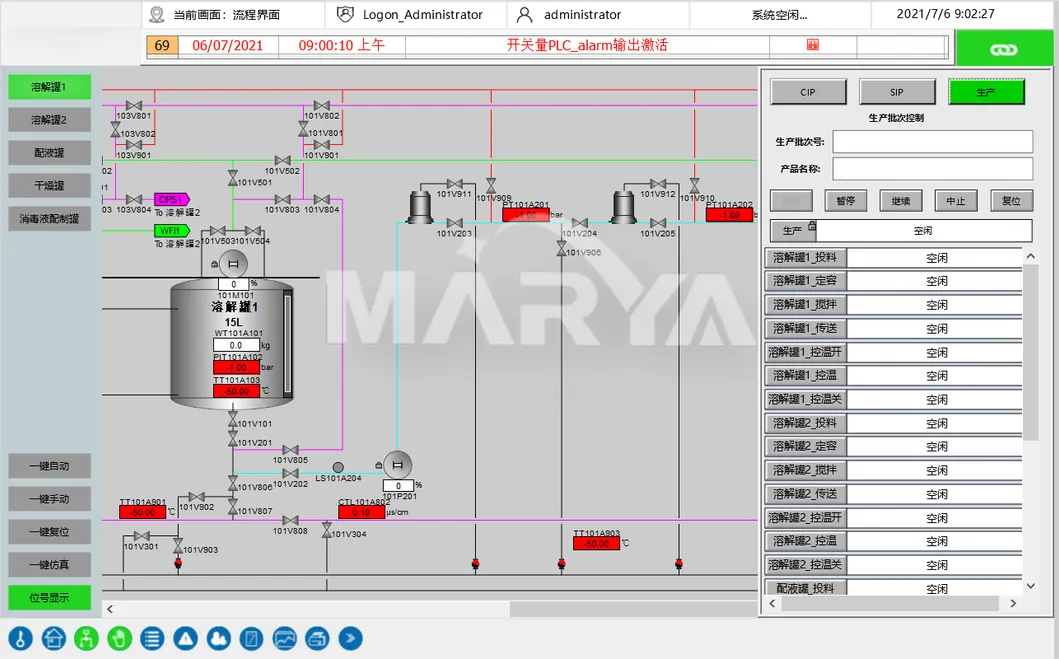
Critical parameter control (temp, DO, pH) with auto-alerts for deviations to ensure drug quality.

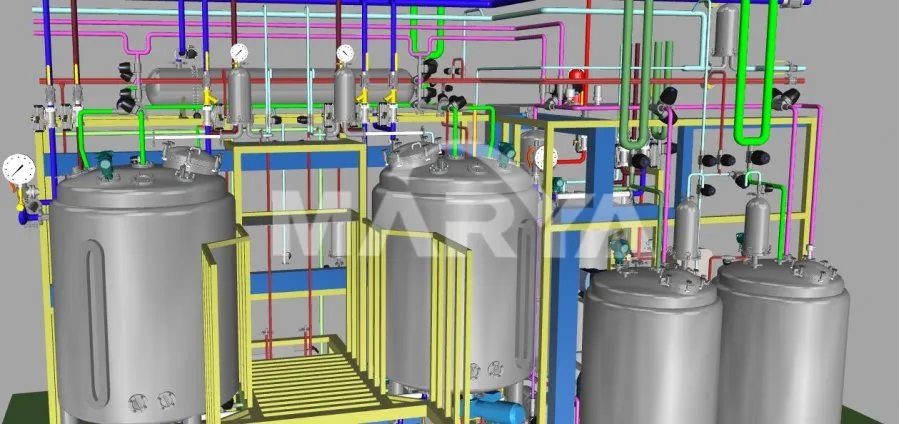
Pre-production 3D modeling optimizes piping, valve, and instrument placement for user-friendly operation.
| Parameter Name | Specifications |
|---|---|
| Working Volume | 2 ~ 15,000L |
| Material | 304, 316L, 904L, TA2 |
| Stirring Type | Upper mechanical, lower magnetic stirring |
| Speed Control Accuracy | 0 ~ 450rpm ±3.0% |
| Temperature Accuracy | -10 ~ 150ºC ±0.2ºC |
| Pressure Accuracy | -0.01Mpa ~ 0.06Mpa ±0.01MPa |
| Weighing Accuracy | 3‰ |
| Protection Levels | IP65 |








The system is highly customizable, with working volumes ranging from a small-scale 2L up to a large-scale 15,000L to meet various production needs.
Our systems are designed to strictly comply with international standards including CE, FDA, GMP, and ISO, ensuring safety and quality for biopharmaceutical production.
We offer both upper mechanical and lower magnetic stirring options, both featuring variable frequency speed regulation for precise control over the mixing intensity.
Yes, the system features fully automated 1-click CIP (Clean-in-Place) and SIP (Sterilize-in-Place) operations with automatic detection of completion.
Tanks are primarily made of 316L stainless steel with electrolytic polishing. Seals are FDA-compliant materials such as EPDM or PTFE to ensure zero contamination.
Absolutely. Integrated SCADA systems provide real-time monitoring and control over temperature, dissolved oxygen (DO), and pH levels with automated deviation alerts.